well so far I think that the addition of Point of Care monitoring has taken away any of the significant benefits of all these agents while not adding in the significant additional drug costs.
Last two times they've found that "one size slips all" (no not a typo) actually doesn't work as well as the drug we already have does right now.
I keep coming back to the observation that all previous problems with warfarin and everything that gives it a bad RAP sheet is down to the simple issue of managing dose.
Wasn't available when President Truman took it but sure is now.
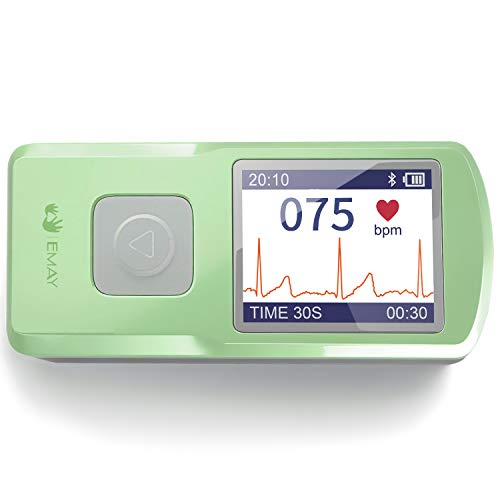
Unlike diabetics who need to test their blood sugar not just dialy but hourly for best effect, for us its weekly.
A finger prick weekly and a small amount of thought once a week and voilla.